|
The cell wall can avoid the cell bursting. As a result, the cell membrane is pushed towards the plant cell wall. When water enters the plant cell, the cell swells up. Hypotonic solutions can cause turgidity in plant cells. The shape of the cell is not changed no swelling or shrinking occurs. Therefore, the cells remain in their normal state. The rates of the movement of water from one solution to the other are equal. When two isotonic solutions are separated from a semipermeable membrane, there is no net movement of solutes across the membrane since there is no concentration gradient between the two solutions. Isotonic solutions have the same amount of solutes per unit volume of solution and the same amount of water. This is due to the equal concentrations of solutes they have. Isotonic solutions are solutions having equal osmotic pressures. Key Terms: Concentration, Hypertonic, Hypotonic, Isotonic, Osmotic Pressure, Solutions, Turgidity 
What is the Difference Between Isotonic Hypotonic and Hypertonic The main difference between isotonic hypotonic and hypertonic solutions is that isotonic solutions are solutions having equal osmotic pressures and hypotonic solutions are solutions having a lower osmotic pressure whereas hypertonic solutions are solutions with a high osmotic pressure.  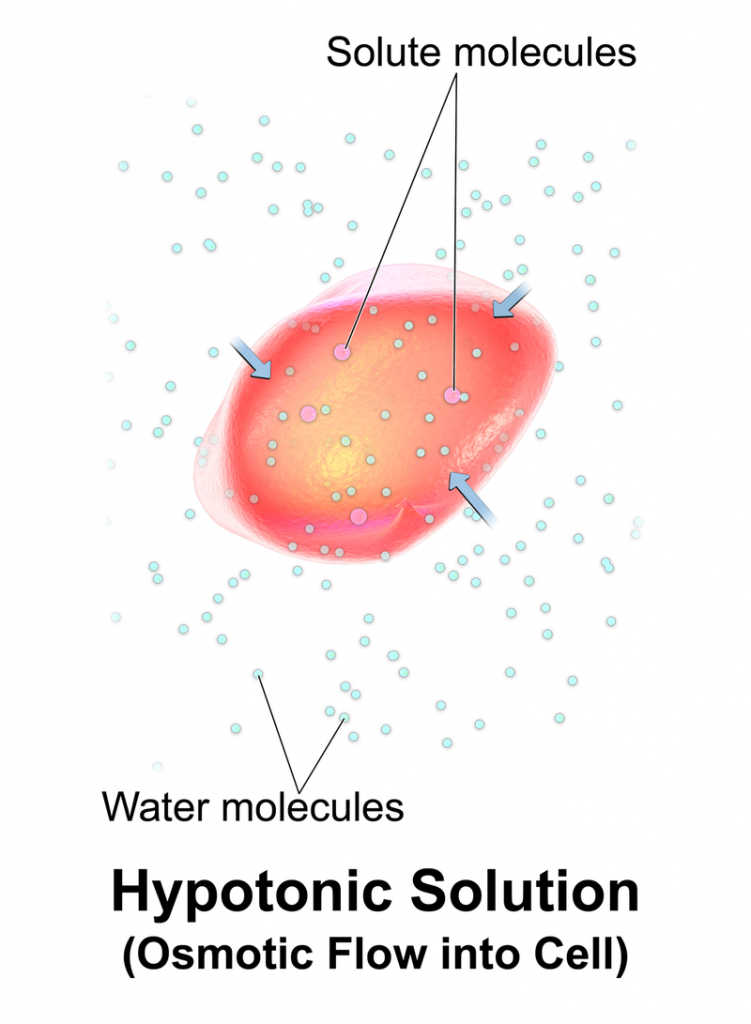
The concentration of a solution determines its osmotic pressure the minimum pressure required to avoid a solution flowing through a semipermeable membrane. The concentration of a solution is the amount of solute present in a unit volume of the solution. There are three types of solutions grouped based on their concentrations. A solution is made by dissolving a solute in a solvent. Main Difference – Isotonic vs Hypotonic vs HypertonicĪ solution is a homogeneous liquid mixture of two or more components.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
